A Game-Changing Approach to Intestinal Transplantation

With a successful intestinal transplant, a soldier with a severe abdominal injury caused by an improvised explosive device can expect to live a nearly normal life. A premature baby whose intestinal wall has been invaded by bacteria can live to see their first birthday.
Intestinal transplants are transformative when they work, but too often they don’t. A first-of-its-kind study now underway at the NC State College of Veterinary Medicine and Duke University could change that.
Liara Gonzalez, CVM assistant professor of gastroenterology and equine surgery, is one of the primary investigators on the study that’s testing a new way to effectively maintain donated intestines and markedly strengthen their viability, lowering transplant failure rates.
The two-year, $1.5 million project is funded by the U.S. Department of Defense through its Congressionally Directed Medical Research Programs office, which awards grants to innovative biomedical research.
“We could absolutely change how people are treated for severe intestinal injury and disease,” said Gonzalez. “It’s extremely exciting.”
Gonzalez is collaborating with a multidisciplinary team from Duke University, including co-primary investigators Debra Sudan, chief of abdominal transplant surgery at the university’s medical school and Katherine Garman, associate professor of gastroenterology. Gonzalez is the only specialist in veterinary medicine on the study’s team.
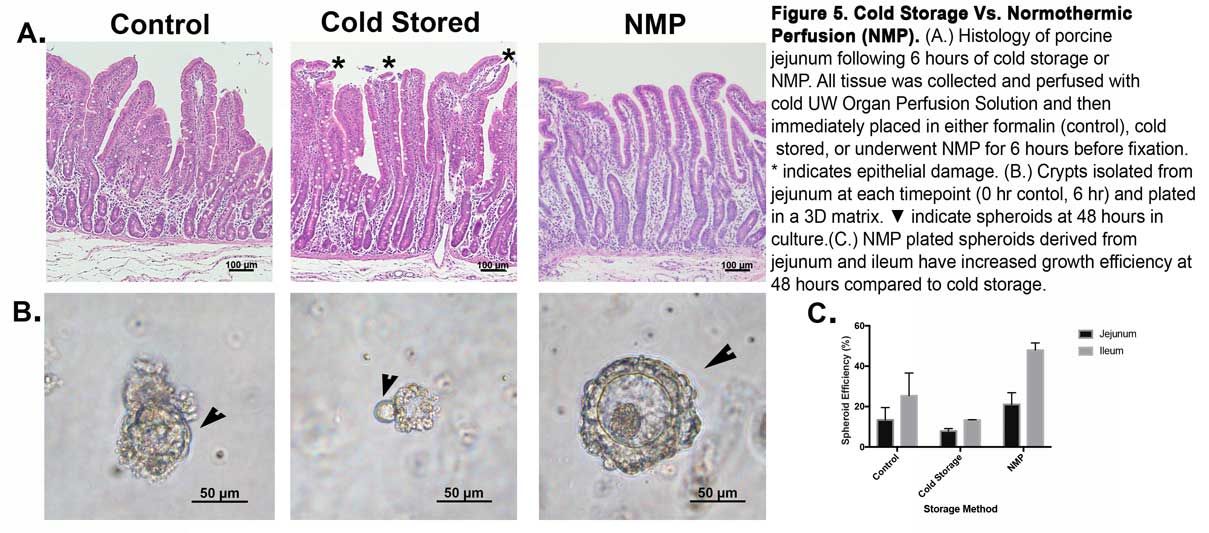
Like other organs used in transplants, intestines are rapidly cooled with a cellular preservation fluid in a bag and then placed on ice. Several studies have connected the length of reduced blood flow, or ischemia, during cold storage with damage to donated intestines, such as decreased epithelial function.
Pre-transplant damage can alter intestinal microbes and the organ’s immune response function. Prolonged cold storage ischemia boosts the risk of complications after transplantation, which can include bacterial infections. Death rates also increase.
The new study will test an alternative method: normothermic machine preservation. In NMP, an organ’s blood vessels are instead perfused with nutrients and oxygen at normal body temperature, preventing ischemia and boosting the chance of a successful transplant. NMP has yet to be used with intestinal transplantation in clinical practice. There are no NMP devices for the intestine currently approved by the U.S. Food and Drug Administration.
“This has huge clinical implications, not just in my lifetime but in the near future,” said Gonzalez. “There are people with traumatic injuries and a lot of people with a large variety of diseases that would benefit from intestinal transplantation.”
Because of damage during preservation, upwards of 87% of intestinal transplant recipients experience acute cellular rejection as soon as 10 days after transplant, the study’s investigators note. Other studies have found that the most serious complications, including organ failure, most often occur within the first 90 days after the transplant.
The NMP method has shown promising results in liver transplantation when compared to cold storage. The method could be used to deliver medications and favorably tweak the immune response of the transplant, which the new study will also investigate.
The study has two main components. The team will test how human intestinal tissue samples obtained through organ donation retain cellular viability and epithelial integrity during both cold storage and NMP. Additionally, an NMP device will be used to preserve and prepare a pig intestine for transplantation into another pig. The pig model is frequently used in such research because its body’s mechanisms, including organs and blood flow, is remarkably close to humans.
“We could absolutely change how people are treated for severe intestinal injury and disease.” – Liara Gonzalez
Intestinal transplantation is performed far less commonly than other organs. According to the United Network for Organ Sharing, 104 intestine transplants took place in the United States in 2018, compared to nearly 22,000 kidney transplants during the same period. Intestine transplants can be performed either with a liver transplant or part of a multi-organ transplant, including combining it with other organs, such as the liver and pancreas.
From the 1990s, when intestinal transplantation first became clinically viable, through July 2019, there have been just over 4,000 intestine transplants completed worldwide, according to the Intestinal Transplant Registry.
The relatively low numbers reflect the primary use of intestinal transplantation as a last-resort, lifesaving measure. Gonzalez said that because of the risks associated with such transplants, the preferred treatment is parenteral nutrition, which intravenously provides necessary proteins, carbohydrates, vitamins and minerals, replacing the intestine’s natural nutrient absorption.
People with intestinal injury can live on total or partial parenteral nutrition, but the treatment is also associated with poor quality of life, high infection risks and emotional disorders.
“People can live, but their lives won’t really be very normal,” said Gonzalez. “ They have to sleep attached to a pump that gives them their food and nutrients for 12 hours into a vein.”
Strengthening the viability of intestinal transplantation has implications for military injuries, which hits home for Gonzalez since her brother, father and grandfather served in the U.S. Navy, but also will impact pediatric medicine.
Infants with necrotizing enterocolitis, a bacterial infection that causes intestinal inflammation and can ultimately destroy the intestinal wall, are often left with short bowel syndrome, when the intestine is not long enough to absorb nutrients. Adults with severe Crohn’s disease, which debilitatingly inflames the lining of the digestive tract, can also develop short bowel syndrome.
“The people who have received successful transplantation — they have a whole new life,” said Gonzalez.
At the CVM, Gonzalez’ clinical and research work focuses on intestinal disease, with a particular goal of preventing equine colic. Her innovative work has long had implications for human health. In 2017, her Intestinal Regenerative Medicine lab developed the most complete recreation of the innermost layer of a horse’s intestine using stem cells. The work helps refine the understanding of tissue damage, aiding the development of effective treatments for gastrointestinal disease.
Earlier this year, Gonzalez and colleagues from NC State and UNC-Chapel Hill published a study that revealed how the body uses reserve stem cells to recover from severe intestinal injury caused by a blockage to the organ’s blood supply. Gonzalez connected with Garman and Sudan after she was invited to speak about her work at Duke two years ago.
“It’s inspiring to be part of this research that will be so high-impact and so beneficial,” said Gonzalez. “We’re ready to go.”
~Jordan Bartel/NC State Veterinary Medicine