Intestinal Pathogens Research
About Our Research
The primary research focus of the Gookin laboratory is to understand how intestinal and gallbladder epithelium arbitrate host defense with an emphasis on minimally-invasive pathogens of comparative relevance to humans and food/companion animals. Our laboratory is currently engaged in research on three of the most prevalent pathogenic causes of diarrhea in the developing world and veterinary medicine including Cryptosporidium parvum, enteropathogenic E. coli, and Tritrichomonas foetus infection. For the past 10 years our laboratory has ardently focused on understanding the pathogenesis of a newly emergent, prevalent, and deadly dysfunction of the gallbladder epithelium of dogs referred to as mucocele formation.
Contribution to Science
Leader in research on pathogenesis and underlying cause of gallbladder mucocele formation in dogs.
Emerging only ~15 years ago, gallbladder mucocele formation is now the most common biliary disease of dogs and its cause is unknown. Our contribution here is mechanistic research investigating the pathogenesis of this unique disease including definitive studies on the histopathologic, microbiologic, proteomic, transcriptomic, and ion transport alterations unique to the epithelial dysfunction, characterization of the metabolic and endocrine disruption occurring in these dogs, clinical trials to alter the course of disease progression, and most recently discovery of environmental exposures unique to this population.
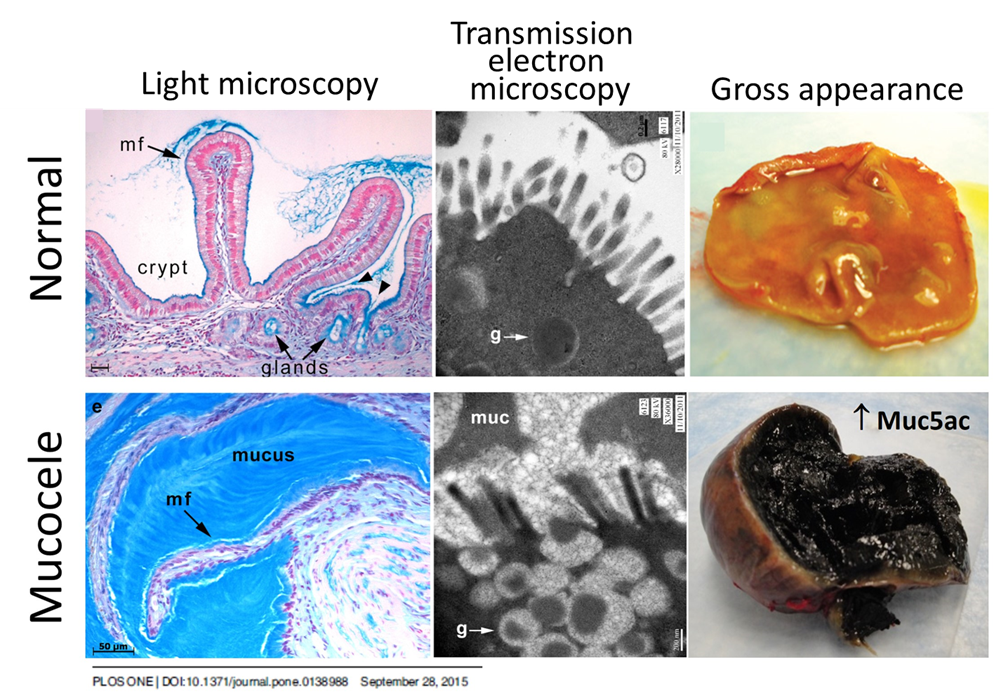

Read more about gallbladder mucocele formation here
Discovery of the protozoal pathogen Tritrichomonas foetus as a new and most common and worldwide infectious cause of colitis in the domestic cat.
Our contribution here is a 23 year track record with over 27 original research publications ranging from discovery of this infection, identification of the causative species, proof of Koch’s postulates of disease causation, establishing prevalence and risk factors, development of methods for diagnosis, conducting basic science studies of disease pathogenesis, and identification of the only known treatment. For these achievements, I received an award of recognition by the American Veterinary Medical Foundation for contribution to advancement of feline medicine.


Read more about gallbladder mucocele formation here
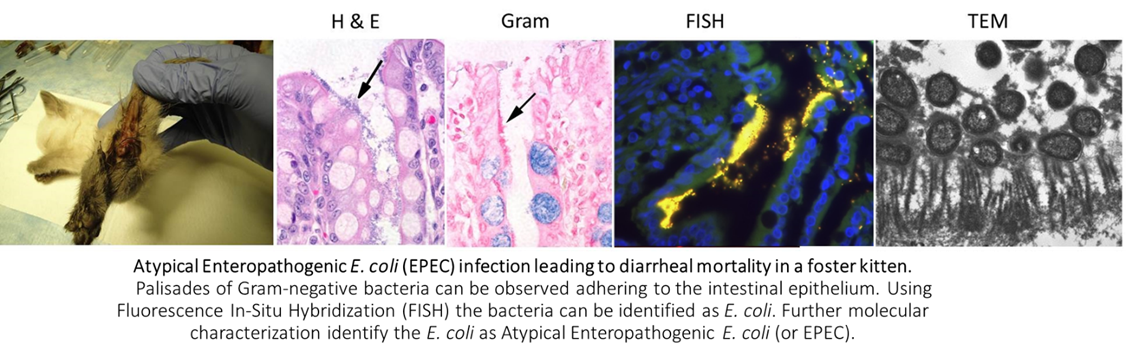
Identification of enteropathogenic E. coli (EPEC) as risk factor for diarrheal death among kittens in the U.S. – a novel model for insight into childhood infection.
Among infectious causes of diarrhea, EPEC carry the highest hazard of death in children less than five years of age in developing countries. While in vitro cell culture models of EPEC demonstrate epithelial dysfunction, translating these findings into in vivo disease mechanisms is a major hurdle due to lack of a good animal model. In the U.S., approximately 15% of kittens will die before they reach 8-weeks of age. These deaths are frequently associated with clinical signs of diarrhea. In the course of investigating infectious causes of diarrheal death in kittens, we are the first to discover a significant association between mortality and EPEC infection. Our current studies utilizing kittens as an experimental model of EPEC infection are the first to characterize the impact of EPEC on intestinal epithelial absorption and permeability in vivo and have identified kitten EPEC as phylogenomically indistinguishable from childhood EPEC isolates. These ongoing studies are generating unique comparative insight into pathogenesis of the infection with dual benefit for veterinary and human medicine.
Novel insight into host strategy for epithelial defense against Cryptosporidium parvum infection.
Cryptosporidiosis is the second leading cause of diarrheal death of children in developing countries and of diary calves in the United States. Our understanding of diarrhea pathogenesis, and therefore development of life saving treatments, is limited by lack of authentic conventional animal models and reliance on cell culture observations. Our contribution here is 15-years of experience in use of a novel neonatal piglet model of infection that recapitulates clinical disease and for which few laboratories have the resources or expertise to utilize. Our early work determined that luminal L-arginine is transported by C. parvum infected epithelium by systems y+ and B0,+ and converted to nitric oxide by NFkB-induced iNOS. We identified nitric oxide as a key mediator of epithelial defense whose mechanism promotes peroxynitrite-dependent anti-cryptosporidial effects, stimulates cyclooxygenase-dependent prostaglandin synthesis in defense of paracellular permeability and increases restitution. In more recent investigations, we identified an entirely novel mechanism by which the intestinal epithelium maintains barrier function against C. parvum infection by inducing an inhibitor of apoptosis protein (XIAP) that represses caspase-3 dependent shedding of intestinal epithelial cells until they reach the villous tips and are physiologically shed. This led us to a new and substantially different conceptual approach to treatment of C. parvum infection based on targeting crypt cell proliferation to promote clearance of infected enterocytes and villous reconstitution. Our observation that C. parvum represses crypt cell proliferation in vivo strongly supports the latter’s role as a key host defense mechanism. Our laboratory has recently identified a specific role for the novel type III interferon, IFN-l3 in mediating epithelial defense against C. parvum.
Complete List of Published Work in My Bibliography:
https://www.ncbi.nlm.nih.gov/myncbi/1liAz-iBjh9AX/bibliography/public/
Engaging Students
Sponsors
Our research isn’t possible without the support of our sponsors. Thanks to the following agencies and foundations for their support:
- American Kennel Club Canine Health Foundation
- Morris Animal Foundation
- Winn Feline Foundation/EveryCat Foundation
- National Institute of Diabetes and Digestive and Kidney Diseases
- Center for Gastrointestinal Biology and Disease at UNC-Chapel Hill
- North Carolina Translational and Clinical Sciences Institute
Research Team
