Flow Cytometry and Cell Sorting
If you are looking for the Clinical Pathology & Immunology diagnostic testing lab service please click here.
Instrumentation
High Speed Cell Sorter

Cytek Aurora CS
The Cytek Aurora CS provides NCSU and to those outside of the university community with the flexibility to perform a wide range of high-speed cell sorting experiments. Our Aurora CS uses full spectrum flow cytometry and detects the entire fluorochrome emission spectrum. This enables the resolution of fluors that are highly overlapped and cannot be resolved via the conventional route (e.g. APC & AlexaFluor647 can be resolved clearly; even if co-expressed). The cell sorter is enclosed in a Class-II Biosafety cabinet, has 5-lasers (355, 405, 488, 561, and 640 nm), 3 scattering & 64 fluorescent channels, and four nozzle options (70, 85, 100 and 130-micron), which provides us with the unique ability to sort a wide variety of cell types. Please contact us to discuss about your experiment and schedule a sorting.

Analyzers
Agilent NovoCyte Advanteon: We have two instrument available for us: 2-laser system (Blue + Red lasers) and 3-laser system (Blue + Red + Violet lasers), . Can analyze up to 11 or 19 colors and utilizes NovoExpress software for acquisition. For experiment planning use the following links for each configuration: 2-Laser, 3-Laser.
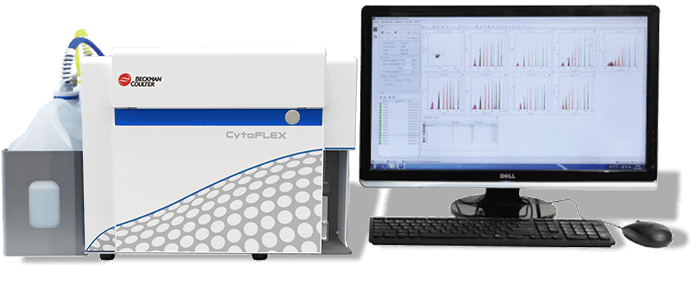
Beckman Coulter CytoFLEX: Has 405 nm (Violet), 488 nm (Blue) and 633 nm (Red) lasers. Supports up to nine parameters and has a vSSC parameter for detection of nanoparticles at higher resolution. The CytoFlex configuration is customizable for specific experiments. The Cytoflex can perform absolute counts. Click here to access instrument configuration.
Gene Expression
NanoString nCounter: The NanoString nCounter Analysis system utilizes novel fluorescent color-coded molecular barcode technology coupled with single molecule imaging to perform digital nucleic acid (RNA & DNA) counting. This technology enables investigators to profile hundreds of targets simultaneously, up to 800 mRNA (or miRNA; which requires an additional step) targets in a single reaction for many kits.

The system consists of a robotic Prep Station for sample processing and the nCounter Digital Analyzer for collecting data.
Automated Cell Counter

LunaII: The LunaII is an automated cell counter that supports brightfield cell counting. It has features to auto-focus, adjust light levels and capture a high-resolution image. You can create custom, cell-specific cell counting protocols or simply use default counting protocol suitable for most cell types.
Scheduling
Aurora Cell Sorter: Scheduling for cell sorting is only available though the flow cytometry core manager. Please contact us to discuss about your experimental needs and schedule a sorting.
Advanteon and CytoFLEX: Scheduling for the Advanteon and CytoFLEX is available through online calendar. Users are given access to the scheduler after completing required introductory and/or instrument training. Please contact the core manager to schedule introductory and analyzer training.
Special Filters
| Excitation | Fluorescence Channels | Fluorochromes |
|---|---|---|
| 488nm | 525/40 BP | Fitc, alexa Fluor 488, CFSE, Fluo-3 |
| 585/42 BP | PE, PI | |
| 610/20 BP | ECD, PE-Texas Red, PE-CF594, PI | |
| 690/50 BP | PC5, PC5.5, PerCP, PerCP-cy5.5, PI | |
| 780/60 BP | PC7 | |
| 638nm | 660/20 BP | APC, Alexa Fluor 647, eFluor 660 |
| 712/25 BP | APC-Alexa Fluor 700, Alexa Fluor 700 | |
| 780/60 BP | APC-Alexa Fluor 750, APC-Cy7, APC-H7, APC-eFluor 780 | |
| 405nm | 450/45 BP | Pacific Blue, V450, eFluor 450, BV421 |
| 525/40 BP | Krome Orange, AmCyan, V500, BV510 | |
| 610/20 BP | Violet610, BV605, Qdot 605 | |
| 660/20 BP | Violet660, BV650, Qdot 655 | |
| 780/60 BP | Violet780, BV785, Qdot 800 |
Services
- Introductory Training
- Instrument Training
- Assisted Acquisition/Analysis
- Cell Sorting
- Analysis Software
- Experiment Design/Consultation
Facility Policies
Facility Access
Users who have been trained for independent operation of the analyzers will be given access to the online calendar system where they can schedule their appointments and will be given 24 hour access to the facility. Please contact core manager to schedule a training and request access to the facility.
Instrument Training
First time users must go through a formal instrument training provided by the flow core. Instrument training by other fellow lab members is not acceptable, and such users will not be granted access to the instruments.
First Time Users
For first time users, it is required to make an appointment with the core manager to discuss your proposed experiment to be sure the core facility has the capabilities to support your experimental needs.
Cancellation
All appointments require 24 hour notice for cancellations. If you do not notify the core manager in writing, you will be charged.
Billing policy
Billing will be based on either the scheduled time or the actual instrument usage time, whichever is greater.
Scheduling Cell Sorting/Assisted Analysis
Scheduling Cell Sorting/Assisted Analysis
Users must schedule cell sorting and other facility-assisted appointments directly with the core manager, Amir Figueroa via email, as far in advance as possible.
Biosafety
The Flow Cytometry facility is a BSL-1 facility. Human (this includes established human cell lines as well as primary cells) and non human primate samples may be run as long as they are fixed on our analyzers. Our cell sorter is considered BSL-2, a Class-II Baker Biosafety hood houses our Aurora CS, which allows the sorting of unfixed cells. If you have questions about this, please contact the core manager or EHS.
Listserv
Users of the facility will automatically be added to a flow cytometry listserv that will keep users up to date with the state of the facility.
Acknowledgments
- Please remember to acknowledge NCSU Flow Cytometry and Cell Sorting facility in all your publications resulting from the use of instrumentation and services at NCSU flow core. Where appropriate, please acknowledge individual members of the Flow Cytometry Core. Following text may be used to acknowledge facility use:
“Flow Cytometry experiments were performed in the Flow Cytometry and Cell Sorting facility at North Carolina State University- College of Veterinary Medicine.”
Rates
| Service | NCSU (per hour) | External Academic (per hour) | External Commercial (per hour) |
|---|---|---|---|
| Aurora CS - Start Up | $ 125 | $ 127 | Contact Us |
| Aurora CS - Sort | $ 215 | $ 217 | Contact Us |
| Advanteon unassisted | $ 59 | $ 63 | $ 73 |
| CytoFLEX unassisted | $ 59 | $ 63 | $ 73 |
| Advanteon assisted (includes staff time) | $ 188 | $ 190 | $ 200 |
| CytoFLEX assisted (includes staff time) | $ 188 | $ 190 | $ 200 |
| Analysis Computer | free | free | free |
| Consultation | $ 71 | $ 76 | $ 81 |
| Seahorse XF Mini- Customer operated | $ 63 | $ 67 | $ 77 |
| Nanostring - Customer Operated (per cartridge) | $ 329 | $ 334 | $ 339 |
| Luna Cell Counter | Contact Us | Contact Us | Contact Us |
| FlowJo License | Contact Us | Not Available | Not Available |
Resources
Flow Cytometry Tutorials
- Introduction to Flow Cytometry (Video Tutorial)
- Introduction to Fluorescence (Video Tutorial)
- Introduction to Flow Cytometry Data Analysis (Video Tutorial)
- Doublet Discrimination Tutorial – Video tutorial that covers different strategies to exclude doublets
- Cyto University: Free to ISAC members
Useful Flow Cytometry Links
- The Flow Cytometry Network: A network to connect with Cytometry community
- BD Biosciences Flow Cytometry Tools
- Purdue University Cytometry Laboratories
- International Society for Advancement of Cytometry (ISAC)
- Biolegend Flow Cytometry Tools
- Optimized Multicolor Immunofluorescence Panels (OMIPs)
- Minimum Information about a Flow Cytometry Experiment
Flow Cytometry Tools
Flow Cytometry Protocols
Flow Cytometry Protocols from Biorad
Tips and Tricks for Designing Flow Cytometry Panels
Training Videos
Fundamentals of Flow Cytometry (Video Tutorial)
Introduction of FACS DIVA Software (Video Tutorial)
Contact Us
Location:
Flow Cytometry and Cell Sorting Facility
CVM Main Building, Room# B303
North Carolina State University – College of Veterinary Medicine
Raleigh, NC 27607
